Also, MOT does not say anything about the geometry and shape of the molecule. $BO =$ $\dfrac$ and so on.Īlso, the MOT uses the concept of bond order to explain the existence of a molecule on the basis of bond order, but this method can neither be feasible nor appropriate to explain about the molecular existence of polyatomic molecules. A pi bond is weaker than a sigma bond, but the combination of pi and sigma bond is stronger than either bond by itself. Quadruple bonds are extremely rare and can be formed only between transition metal atoms, and consist of one sigma bond, two pi bonds and one delta bond. Benzene has molecular formula C 6 H 6 so there is the presence of one double bond in between alternate carbon atoms and each carbon is attached to two carbon atoms by means of a single bond and a double bond and a hydrogen atom by a single bond. Two pi bonds are the maximum that can exist between a given pair of atoms. Therefore, there are 2 bonds present in the. Because these four electrons are in the pi orbitals, the C 2 molecules two bonds will be pi bonds alone, with no sigma bonds. 2 p x and 2 p y, according to Molecular Orbital theory. The Molecular Orbital Theory describes the bond order to be basically the difference between the number of bonding and antibonding electrons, divided by two. The ratio of and bonds in benzene is 4:1. The last two paired sets of electrons enter the degenerate (identical energy) pi-bonding set of orbitals, i.e. Pi bonds are usually weaker than sigma bonds.The C-C double bond, composed of one sigma and one pi bond, has a bond energy less than twice that of a C-C single bond, indicating that the stability added by the pi bond is less than the stability of a sigma bond. The question says that the molecule has two pi and half sigma bond hence the bond order of the molecule will be 2.5 Properties Two p-orbitals forming a -bond. Similarly, we will now check for the number of bonds present in the molecule we are talking about in the question. 
Like if 2 atoms in a molecule are bonded via a single bond, their bond order will be one if via a triple bond. We all know that bond order basically gives us the number of bonds or the number of electron pairs present between two atoms in a molecule. Also, the molecule which has two pi and half sigma bonds will have a bond order of 2.5 This can be illustrated by comparing two types of double bonds, one polar and one. 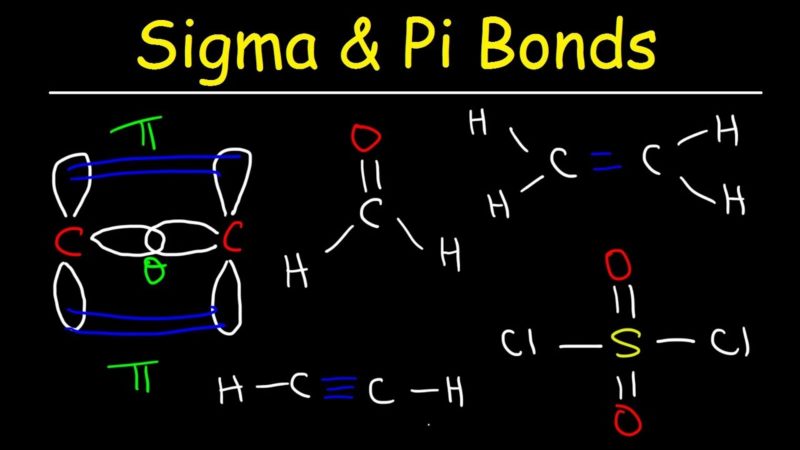
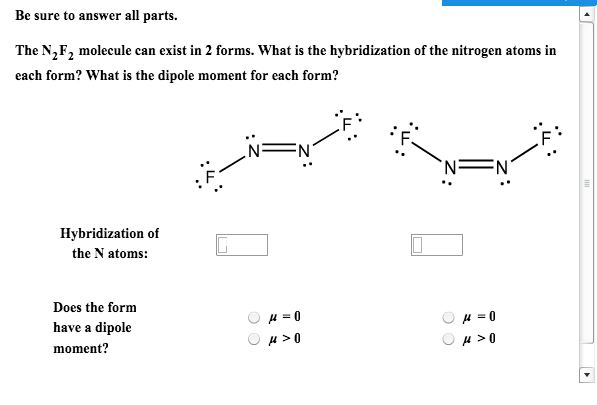
The two bonds in the triple bond are formed from overlap. Give the molecular structure, hybridization, bond angles, and an example for each molecule. Molecule C consists of two bonds and two bonds. Now that we understand the difference between sigma and \(\pi\) electrons, we remember that the \(\pi\) bond is made up of loosely held electrons that form a diffuse cloud which can be easily distorted. The sigma bond is formed from head-to-head overlap of the sp hybrid orbitals from the C and N atoms. Molecule A has a hybridization of sp3 Molecule B has two more effective pairs (electron pairs around the central atom) than molecule A. Accordingly, see how many sigma and pi bonds each of the molecules can form. Mobility Of \(\pi\) Electrons and Unshared Electron Pairs. If you would use that formula for propane, number of sigma bonds will be 3 + 8 - 1 = 10, as shown above.Hint: First write down the electronic configurations of the given molecules in the option and see the number of electrons in their bonding and antibonding orbitals. Number of sigma bonds = number of atoms - 1 (which you can determine using the molecular formula ) Lewis dot symbols are useful for tracking electrons. elements of an ionic compound usually carry the same charge. How many bonds can each atom make without hybridizationa. ionic bonds usually occur between elements with high and low electron affinities. The valence electron configurations of several atoms are shown here. Also, in this particular case ( for saturated aliphatic alkanes ) you can use a formula: ionic bonds are a result of electrostatic forces. It is a saturated compound, so you will only have simple ( sigma ) covalent bonds - there are no pi bonds. a b 15 Work out the number of sigma and pi bonds in each of the following: a O2 g N2F2 b. Let's take the propane extended structure formula (which i stole from the answer above): Bond energies in single and double bonds. 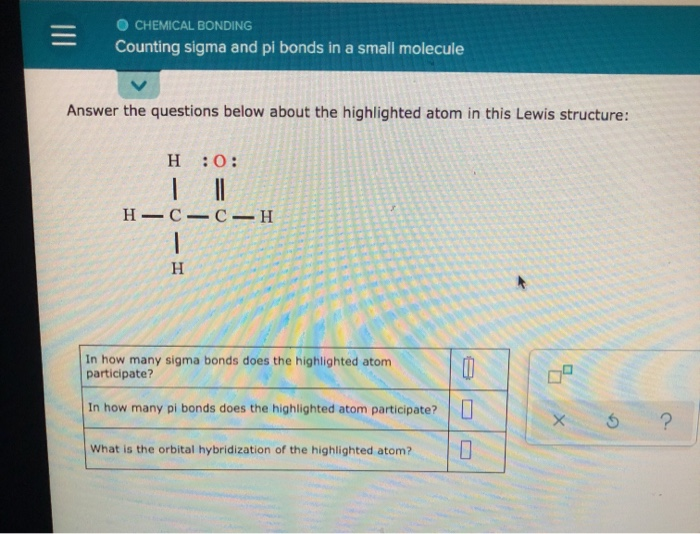
Each simple covalent bond (represented by the line between 2 atoms) will be a sigma bond Īny other different from that ( double, triple covalent bonds ) will still have one sigma bond, the rest of the lines being counted as pi bonds Start off by writing down the respective compund using the extended structure model. Here you have a more in-depth analysis of structure formulas.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
